EXECUTIVE ABSTRACT
Strength of recommendation: Weak in favor of the technology.
Technology: Ranibizumab and bevacizumab.
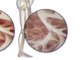
Indication: Age-related macular degeneration.
Characterization of the technology: Ranibizumab and bevacizumab are angiogenesis inhibitors (antibodies) that inhibit the activity of human vascular endothelial growth factor and, consequently, inhibit the formation of new blood vessels preventing therefore the loss of vision in individuals affected by Age-related Macular Degenaration (AMD).
Question: Are ranibizumab and bevacizumab effective and safe for the treatment of age- related macular degeneration?
Search and analysis of scientific evidence: We searched The Cochrane Library, Centre for Reviews and Dissemination, Medline and LILACS databases aiming to find systematic reviews comparing treatment containing ranibizumab or bevacizumab regimens for the treatment of age-related macular degeneration. Health Technology Assessments (HTA) were searched on the websites of national and international agencies.
Summary of results of selected studies: The results presented by the systematic reviews showed that both ranibizumab and bevacizumab are effective when compared to other available therapies for the treatment of AMD. Both drugs prevent the progressive loss of vision and / or improve visual acuity. However, there are no literature studies comparing the effectiveness of ranibizumab versus bevacizumab (head-to-head) to prove the clinical superiority in relation one to each other. The advantage pointed out by studies that focused on economic evaluation is the difference between the cost of these drugs. Ranibizumab in all these scenarios is more expensive and to make it cost-effective it is necessary that its price decreases. Bevacizumab has equivalent efficacy and lower cost which has promoted discussion regarding its off-label2 use.
Recommendations: We recommend the use of ranibizumab or bevacizumab for the wet form of age-related macular degeneration in patients aged over 50 years.
Full content in Portuguese















Adicionar Comentário