EXECUTIVE ABSTRACT
Strength of recommendations: Weak in favor of the technology.
Technology: Temozolomide.
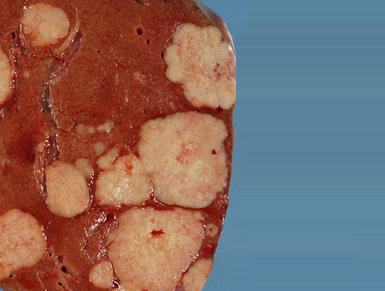
Indication: Temozolomide is indicated for treatment of patients with recurrent malignant glioma, such as glioblastoma multiforme or anaplastic astrocytoma (astrocytoma high-grade malignancy), who have already received chemotherapy or in those in whom conventional chemotherapy was not effective.
Characterization of technology: Temozolomide is a antineoplastic alkylating agent. These agents add an alkyl group to various electronegative groups of cellular DNA and thus alter or prevent cell replication interfering with tumor growth.
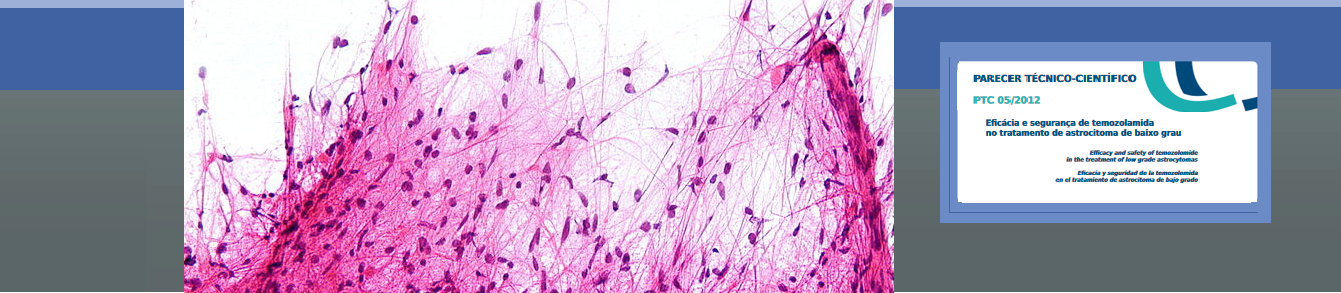
Question: Is the use of temozolomide effective and safe as a treatment alone or adjunctive Low Grade Astrocytoma?
Search and analysis of scientific evidence: We searched The Cochrane Library (via Bireme), Centre for Reviews and Dissemination (CRD), Medline via PubMed, and Clinical Trials LILACS databases aiming to find systematic reviews (SR) of clinical trials comparing temozolomide with other therapeutic options for the treatment of low grade astrocytoma. Health Technology Assessments (HTA) were searched on the websites of national and international agencies. We selected studies published in all languages.
Summary of the results of the selected studies: After full reading two systematic reviews (SR) were selected and four clinical trials. The selected studies were classified as low quality evidence. The first SR used meta-analysis and assessed the efficacy of three different dosing schedules of temozolomide to treat Low Grade Gliomas (LGG) and including 18 phase II trials. This review did not answer our question completely and presented no clinically significant evidence. The second systematic review analyzed qualitatively phase II studies to create a guideline for prescribing and use of temozolomide in the treatment of brain tumors. In this RS, the grade II gliomas were addressed by five studies that evaluated the efficacy and/or tolerance of temozolomide. The question was well structured, although unspecific – the authors sought evidence of the use of temozolomide in different contexts: duration of treatment, rare tumors, drug combinations and directions and doses off-label2 cases. Only two studies showed evidence level B2. The included studies presented inconsistent results that corroborated the use of temozolomide in LGG after failure of radiation therapy and as first-line treatment for more extensive gliomas. The four included randomized clinical trials are still ongoing and aim to evaluate the use of temozolomide alone or as adjuvant compared to radiotherapy – the current standard treatment.
Recommendations: Up to this date there is no available evidence to support the use of temozolomide for low-grade gliomas (Grade I and II) between then astrocytomas (Grade I and II). The results of ongoing clinical trials will be essential to define the best treatment strategy for this health condition.
Full content in Portuguese















Adicionar Comentário